|
GSL’s SnapGene desktop software ( ) is used in more than fifty countries, in virtually every major research institution, and in most of the largest pharmaceutical and biotech companies worldwide. GSL Biotech was founded by scientists and software designers to meet the everyday needs of molecular biologists. The companies have stated this is a first level integration, with a deeper integration in development. This integration takes advantage of the strengths of both companies while giving common customers significant added flexibility and convenience. All changes to any file will be recorded and stored in the LabArchives platform, including automatic full versioning. Users can open SnapGene files from their notebooks within LabArchives. Now, SnapGene users can upload sequence files directly to their LabArchives notebook. Among the data which can be stored in LabArchives are cloning protocols and plasmid sequences. SnapGene specializes in the visualization and simulation of molecular biology procedures and LabArchives enables researchers to store all of their research data records in a secure, compliant online platform. (PRWEB) JGSL Biotech has released a new integration to enable SnapGene software to connect seamlessly with LabArchives, the leading cloud-based Electronic Lab Notebook (ELN) platform. Scientists can now use SnapGene software together with LabArchives ELNĬHICAGO, ILL AND CARLSBAD, CALIF. If you want to design primers for other applications where efficiency and specificity of the primers can be taken into account, you shouldn't use SnapGene, use CLC Main Workbench or Primer-Blast instead.SnapGene Releases New Integration with LabArchives If you are happy with the sequence of the primer click Add primer to template. Select to make a primer for the Bottom strand: To transform the selection into a primer expand Primers in the top menu and select Add primer. Select a sequence that starts after the stop codon of Rab5 with the correct Tm: Try to do the exercise without peeking at the solution. So the only requirements of the reverse primer are its location (directly after the stop codon) and its Tm (60-62☌). This means we can use the existing BamH1 site right downstream of the rab5 gene. If you are happy with the sequence of the primer click Add primer to template (green).įor cloning Rab5 into the vector later on, we will use a BamHI restriction site. To do this click at the 5’ end of the primer and type a random sequence of 6 nt (red): To shield the site we will some random nucleotides to the 5' end of the primer. The sequence of the site is now added to the 5' end of the primer (blue). Select BspE1' in the site box (red) and click the Insert button (green): To add the restriction site select the Insertions tab: You can just type the sequence you want to add.You can insert codons, restriction sites and tags.There are two ways to add a sequence to the primer: To clone the gene you need a BspE1 site at the 5' end of the primer. Select to make a primer for the Top strand: As you elongate your selection, SnapGene automatically displays the length and Tm of the selection: Go back to the Sequence view of mRFP1-Rab5 and select a sequence that starts with the ATG start codon of Rab5. The only thing that is still variable in this case is the length of the primer, which will be determined by the Tm that we wish. We want primers with a Tm in the range of 60-62☌. 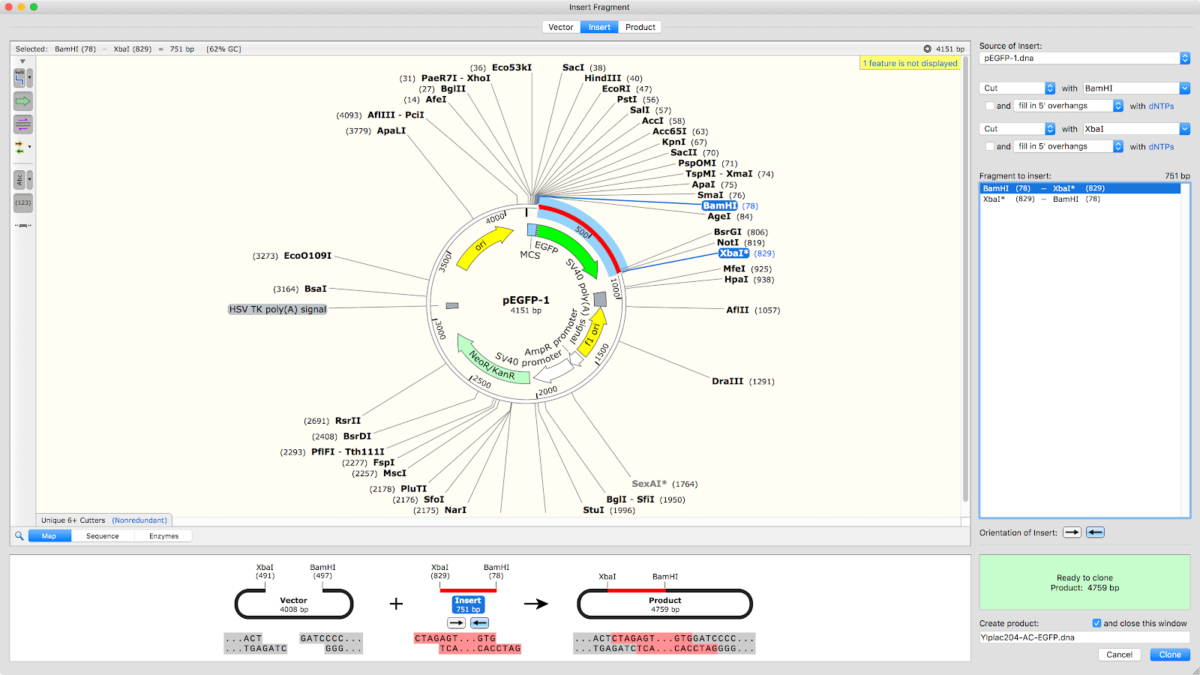
The forward primer should also contain a BspE1 restriction site to fuse the gene to AcGFP1. For this, our choice of primers is restricted: the forward primer needs to start with the Rab5 start codon so its location is fixed. 
For cloning this is often the case.įor instance, we want to make primers to clone the Rab5 gene from plasmid mRFP1-Rab5 as a fusion to the AcGFP1 gene in plasmid pAcGFP1-C1. Only when you have no flexibility in your choice of primers, I would use SnapGene to design them.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
